Year: 2013 Vol. 79 Ed. 6 - (5º)
Artigo Original
Pages: 673 to 680
Correlation between symptoms and course duration of upper aerodigestive tract cancer at early and advanced stages
Author(s): Francis Balduino Guimarães Santos1; Jose Jacinto Branco Vasconcelos-Raposo2; Maria do Carmo Tolentino Figueiredo3
DOI: 10.5935/1808-8694.20130125
Keywords: clinical course; late diagnosis; neoplasms of the head and neck; primary prevention; signs and symptoms.
Abstract:
There still are many clinical and biological aspects of the natural history of cancer of the upper aerodigestive tract to be unveiled; which in Brazil is a direct consequence of the failure of systematic prevention and early diagnosis campaigns.
OBJECTIVE: To analyze the signs and symptoms presented by patients with the disease at initial and advanced stages. Other variables such as disease duration, general and nutritional status were considered.
METHOD: A historical cohort study with a cross-section involving 895 subjects with cancer of the upper aerodigestive tract.
RESULTS: Clinical findings were not statistically correlated with disease progression, nor with the disease in early stages, but it showed rapid disease development.
CONCLUSION: The results suggest a disease of insidious onset in the early stages and fast course afterwards. The long disease duration - greater than three months, was associated with worsening in general and nutritional states of patients.
![]()
INTRODUCTION
Head and neck cancer is the sixth most prevalent primary malignant neoplasm in the world, accounting for 5% of all cancers in men and 2.5 % in women1, being epidemiologically important in many countries worldwide, including Brazil2,3.
The most affected anatomical site by head and neck cancer in the upper aerodigestive tract, distributed among the oral cavity (35%-40%), oropharynx (30%), larynx (25%) and hypopharynx (7%)4. The most prevalent histological type of cancer located in the upper aerodigestive tract is the Squamous Cell Carcinoma (SCC) in 90% of the cases, and its main etiological factors are tobacco smoking and alcoholic beverages drinking5-7, although infection by the human papilloma virus (HPV) has been responsible for at least 10% to 30% of oropharyngeal cancers8.
Clinical staging at the time of diagnosis is the most important prognostic factor affecting survival, but it is advanced around 75% of cases, especially in developing countries. As a result, about 40% to 60% of patients with SCC have locoregional recurrence and 20% to 30% progress with distant metastases8. After treatment, the overall mortality rate stands at around 40% to 60% in developed countries, with a global average around 46%9. The reasons described in the literature to justify this high incidence in advanced phases are distributed in a miscellany of situations: cultural and economic factors, poor social support, lifestyles, demographics and geographic factors, as well as difficulties in having access to healthcare10-13. There is no consensus in the literature regarding barriers to the early diagnosis of SCC.
Studies in Brazil show that the population is unaware of the risk factors associated with these cancers14,15. This is partially due to low levels of schooling and low household per capita income16,17. Another study suggests that the early diagnosis of SCC may be hampered by the fact that the initial lesions, with few symptoms, are not appreciated either by the individual or by healthcare workers12. They also suggest that these symptoms can be confused with other common diseases affecting the population. More so when these symptoms have never been presented to them as cancer harbingers18,19. Modern health thinking is based on the co-responsibility of the individual for his own health20.
In Brazil, there are selective campaign against oral cavity or laryngeal SCC and most of them have been highly ineffective, suggesting the need for further understanding the biological and epidemiological processes behind these types of cancer15,16,21, hence the finding of a carcinogenic lesion remains the most effective clinical intervention22,23. In terms of research, much emphasis has been given to the risk factors and their causal relationships, and less has been done regarding its natural clinical history.
Finally, the disease remains challenging because of its diagnoses in advanced stages, its high morbimortality24 and hence the negative impact on budgets. There are very few correlational studies between the various symptoms, clinical staging and disease course vis-à-vis the initial diagnosis, since it may help to understand human behavior in different regions of the world and outline an epidemiological profile with specific health actions concerning the population exposed to certain risk factors.
SCCs may be considered as a separate disorder in the four anatomical sites and traditionally focused on the specialist's view or be presented as a single disease25,26. In this approach, there is a strong increase in its incidence, taking into account similar epidemiological aspects, and there may be a convergence of combined health actions with cost reduction - vision focused on public health. With both approaches, the present study has the "overall objective" to describe the sociodemographic, behavioral, and clinical characteristics of the sample. The "secondary objectives" are to associate disease course with its staging in initial and advances stages, as well as establish the temporal relationship of the signs/symptoms with the patients' general and nutritional status at the time of diagnosis.
METHOD
Historical Cohort Cross-Sectional Study - it follows the same characteristics of the longitudinal cohort study, however establishing a restricted time point (cut) to assess the elements studied, with no follow-up over time. For the study we selected 895 patients with SCC, between March 1996 and January 2009, seen at the head and neck surgery clinics located in Montes Claros/Minas Gerais - Brazil. We used standard patient charts from only one specialist. This study was approved (Resolution 131 - CEPEX/2008, Ethics Board under protocol # 005/2008) and did not receive funding for its execution. The variables used are described below.
Sociodemographic variables (gender, marital status, age group, place of residence and occupation) and lifestyle habits harmful to health, such as smoking and drinking alcoholic beverages, in addition to the following clinical variables: anatomical sites and subsites according to the International Classification of Diseases (ICD, 10th ed., 2000)27 as follows: oral cavity, oropharynx, larynx and hypopharynx. The criteria used for staging followed the TNM system of the International Union Against Cancer (IUAC TNM, 6th ed., 2002)28. Patients with diagnosis dates prior to 2002 had their staging updated under current TNM standards. Disease progression was considered as reported by the patient, between the dates of the first symptoms to the first visit to a specialist in head and neck surgery (short: < 3 months; long:>3 months). Nutritional status was assessed according to a subjective global assessment of the nutritional state29,30 considering the patient's report of weight loss, current weight of the patient, the medical and physical characteristics presented by the patient or by the association of this information. The general health was assessed by the clinical classification from the Zubrod-Karnofsk31 Performance Status assessment.
We calculated the frequencies, mean values and odds ratios for possible associations between the variables investigated. We used the Statistical Package for Social Sciences (SPSS V. 19®). Initially, we performed a descriptive analysis of the data. Subsequently, due to the nature of the variables, we used the nonparametric tests, a bivariate analysis (χ2 test, Mann-Whitney test) and multivariate data analysis (binary logistic regression). The significance level of 5% was retained for the statistical procedures performed (p = 0.05*).
RESULTS
The conditions most frequently encountered were: males (84.8 %) in the ratio of 5.6 ♂ : 1 ♀. Age ranged from 7 to 92 years, with a mean value of 59.6 years and incidence peaks between the 5th (28.6 %) and 6th (24.8 %) decades of life. We found a higher number of smokers/former smokers (91.5 %) and alcoholic beverages drinkers/former drinkers (86.4 %). As for skin color, the distribution was as follows: whites 227 (30.6 %), browns 417 (56.1 %) and blacks 99 (13.3%). Regarding marital status, married subjects represented 64.8 % (449), singles 17.3% (120), widow (er) 12 % (83), separated 4% (28) and 1.9% others (13).
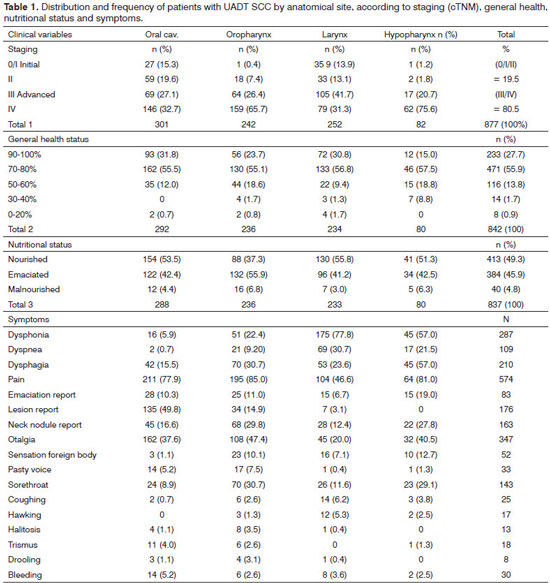
The anatomical sites affected were distributed as follows: oral cavity (33.9%), larynx (28.8%), oropharynx (27.9%), and hypopharynx (9.4%) - Table 1. On this table one can also notice the most affected subsites. Considering all the anatomical sites, clinical staging upon diagnosis was advanced in 80.5% of patients. The anatomic site at which the disease was most advanced in stages III/IV was the hypopharynx with 96.3 % - Table 1.
Considering the nutritional status, 52.1 % of the patients were emaciated or malnourished upon diagnosis and, as far as general condition is concerned, 55.9 % had mild to moderate impact - Table 1.
Symptoms were observed in two ways: separately, for each anatomical site and together - SCC being considered here as a single disease. In the former, and as expected, the most frequent symptoms were distributed in different ways for each anatomic site. The three main symptoms in order of frequency for the oral cavity were: pain, lesion reported by the patient and earache. The oropharynx had the following distribution of symptoms: pain, dysphagia and odynophagia. For the larynx, they were: dysphonia, pain and dyspnea, and for the hypopharynx: pain, dysphagia and dysphonia.
Otherwise, upon considering all anatomical sites together, some differences are noteworthy and the most prevalent symptom of the sample is represented by tumor pain (71.5 %); followed by reflex otalgia, dysphonia, dysphagia, reporting the lesion - the patient reporting a neck lump and dyspnea - Table 1.
After considering the differences in clinical behavior (signs and symptoms) of UADT SCC obtained by separate and joint evaluation of anatomical sites, one can notice their association with the initial and advanced staging.
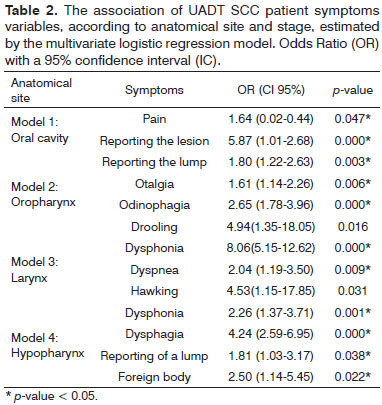
Considering again the anatomic sites separately through a multivariate logistic regression between staging, anatomical site and symptoms, the results show statistically significant associations for advanced disease. They were statistically significant in the oral cavity, pain and injury reported by the patient; in the oropharynx, the reporting of a lump by the patient, otalgia and drooling; in the larynx: dysphonia, hawking and dyspnea; and in the hypopharynx: dysphonia, dysphagia, patient reporting a lump and a foreign body sensation - Table 2.
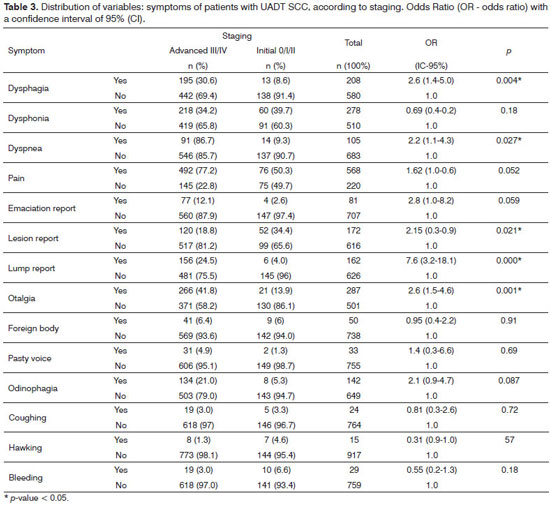
Considering now the anatomical sites together, through the distribution of symptoms according to staging, we obtained statistically significant associations between symptoms and advanced stage, these being: the report of the nodule, otalgia, dysphagia, dyspnea and the account of a lesion - Table 3.
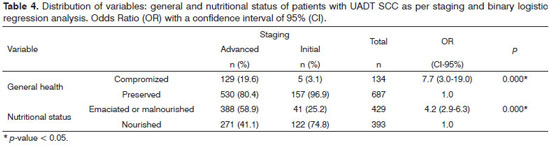
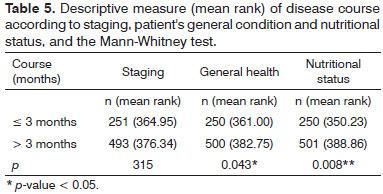
We found statistically significant associations between staging in advanced stages of the disease with the general and nutritional status variables - Table 4.
We found no association between disease course and its staging. There was no statistically significant difference between disease course and the initial or advanced stages. Unlike hypothesized (H1), the disease course did not correlate with staging; however, the long disease course (over three months) was correlated with worsening nutritional states and general health of the patients - Table 5. The time of complaints reported by patients (n = 778) ranged from 1 to 60 months (30 to 1,800 days), with an average of 150 days (SD = 235.44).
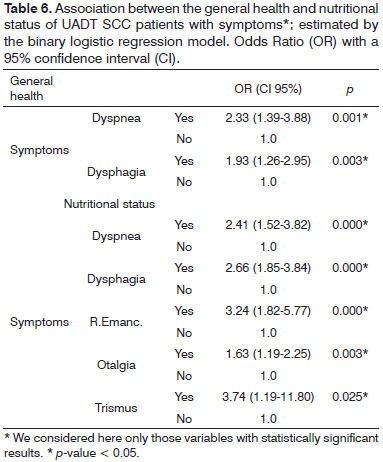
One can observe the variables that correlate with worsening patient nutritional status, such as dyspnea, dysphagia, weight loss reported by the patient, otalgia and trismus. Similarly, the nutritional status variable correlated with general health worsening. Now, 'dysphagia and dyspnoea' were statistically significant vis-à-vis the worsening in their general health - Table 6.
DISCUSSION
In this study, the epidemiological profile of the UADT SCC patients was similar to findings published in the literature32-34. The significant findings regarding gender 5.6 ♂ : 1 ♀ , are partly justified by sociocultural factors; men being more exposed to carcinogenic agents for the development of UADT SCC (risk factors: tobacco and alcohol). The advanced stage found in 80.5% of patients did not differ from the literature33,34 but confirms the seriousness regarding the epidemiology of UADT SCC.
Pain, in its various forms, was the most prevalent general symptom, affecting 71.5 % of patients with UADT SCC, possibly due to the high number of patients with advanced disease, similar to that of another study35. It is known that pain increases the degree of suffering of patients, with significant reduction in hours of sleep, leading to drops in productive capacity, absenteeism and money spent with medications, which ended in a Via Crucis for social support and medical care. This fact shows the importance of pain as a recognition factor of the state of sickness of UADT SCC patients19, but this, unfortunately, already predicts advanced disease. Such as pain, many signs and symptoms are associated with advanced disease in a statistically significant fashion, which does not favor prognosis.
The UADT SCC, which was evaluated by two approaches, as different diseases or as a single disease, presented somewhat similar information. The former had heterogeneous symptoms, which is unevenly distributed among the four anatomical sites, but did not show any pathognomonic sign or symptom to either location. Although the anatomical sites are distinct and have different functions together, their anatomical proximity can make large tumors involve more than one anatomical site or subsite simultaneously, thus producing mixed symptoms. The symptoms listed here were associated with advanced disease. The UADT SCC evaluated as a single disease had symptoms associated with advanced disease such as reporting of the lump, dysphagia, reports of a lesion and dyspnea.
It is difficult to understand why UADT SCC is not diagnosed at an initial stage, but the various symptoms were mostly associated to an advanced stage. Possibly, the initial symptoms are subtle, disregarded by the patient and the disease evolves progressively and rapidly to the advanced stage, not yielding to an initial diagnosis in proper time19.
However, the population has no knowledge or clinical conditions to identify initial or precancerous UADT SCC lesions12,19,23, a finding essential to prevention. The active search for precursor UADT SCC lesions (screening) should be reconsidered for individuals with risk factors for its development (over 45 years old, smoker and/or drinker), as already observed in other studies with favorable results35-37. There is a range of medical equipment currently available to perform imaging of the upper aerodigestive tract (laryngoscopy), which can be offered to the community by the public healthcare system; however, at a cost to be assessed. Primary and secondary care require investments in physical infrastructure and technology for proper action in the prevention and early diagnosis of UADT SCC, as it was done for cervix cancer38.
Some symptoms were significantly associated with the physical decline of patients, such as dyspnea, dysphagia and weight loss. Individuals with mechanical constraints for breathing and/or feeding, due to tumor invasion, have their nutritional status compromised39. These symptoms may be the ones most associated with the wasting state of the disease and can have an unfavorable impact on treatment and prognosis.
The disease course between the first signs and symptoms until the patient's motivation to seek medical care was long in this series (patient's delay), around 150 days, but also comparable to results published in the literature40. The long disease course (> three months) correlated with worsening nutritional and general health status, similarly to that found by other studies, require these patients to receive greater attention in terms of clinical ad nutritional support41,42.
CONCLUSION
Among the results, two were really striking when considering UADT SCC as a single disease: (1) there were no statistically significant signs and symptoms of early UADT SCC, and (2) there was no correlation between the disease course and its staging (patient's delay). This proved, and as the most important conclusion, that when the symptoms become noticeable to patients, they are already an indication of an abnormality, in fact already foreshadowing advanced disease, suggesting rapid evolution of early disease stage to more advanced stages.
The long course of UADT SCC - more than three months, can be regarded as a predictor of worsening in the patient's general and nutritional status. Similarly and specifically, symptoms of "dyspnea and dysphagia" joined the overall status of these patients.
CONFLICTS Of INTEREST
The authors declare no conflicts of interest.
REFERENCES
1. Curado MP. S35 The Brazilian experience on mass screening. Oral Oncol Supp. 2007;2(1):123.
2. Boyle P, Levin B. World Cancer Report 2008. Lyon: World Health Organization; 2009. p.13-5.
3. Instituto Nacional do Câncer: Incidência de Câncer no Brasil: Estimativas 2012. [Acesso em 20 Dezembro de 2012]. Disponível em: http://www.inca.gov.br/estimativa/2012.
4. Barbosa MM. Diagnóstico e tratamento dos tumores de cabeça e pescoço. 1ª ed. São Paulo: Atheneu; 2001. p.491-5.
5. Biazevic MGH, Castellanos RA, Antunes JLF, Michel-Crosato E. Tendências de mortalidade por câncer de boca e orofaringe no Município de São Paulo, Brasil, 1980/2002. Cad Saúde Pública. 2006;22(10):8-15.
6. Santos RA, Portugal FB, Felix JD, Santos PMO, Siqueira MM. Avaliação epidemiológica de pacientes com câncer no trato aerodigestivo superior: relevância dos fatores de risco álcool e tabaco. Rev Bras Cancerol. 2012;58(1):21-9.
7. Avi ALRO, Tanimoto HM, Queiroz CDS, Gerim LR, Zuza EP, Trevisani DM, et al. Neoplasia de boca e orofaringe: um estudo transversal na Fundação Pio XII - Hospital do Câncer de Barretos. Rev Odontol UNESP. 2012;41(4):273-80.
8. Siu LL. Preface: head and neck cancer. Hematol Oncol Clin North Am. 2008;22(6):XIII-XV.
9. Gervásio OL, Dutra RA, Tartaglia SM, Vasconcellos WA, Barbosa AA, Aguiar MC. Oral squamous cell carcinoma: a retrospective study of 740 cases in a Brazilian population. Braz Dent J. 2001;12(1):57-61.
10. Onizawa K, Nishihara K, Yamagata K, Yusa H, Yanagawa T, Yoshida H. Factors associated with diagnostic delay of oral squamous cell carcinoma. Oral Oncol. 2003;39(8):781- 8. PMID: 13679201 DOI: http://dx.doi.org/10.1016/S1368-8375(03)00075-7
11. Kowalski LP. Causes of diagnostic delay of oral cancer. Oral Oncol Supp. 2007;2(1):123-3.
12. Tromp DM, Brouha XD, Hordijk GJ, Winnubst JA, de Leeuw RJ. Patient and tumour factors associated with advanced carcinomas of the head and neck. Oral Oncol. 2005;41(3):313-9. PMID: 15743694 DOI: http://dx.doi.org/10.1016/j.oraloncology.2004.09.008
13. Guidry JJ, Greisinger A, Aday LA, Winn RJ, Vernon S, Throckmorton TA. Barriers to cancer treatment: a review of published research. Oncol Nurs Forum. 1996;23(9):1393-8. PMID: 8899755
14. Antunes JLF, Toporcov TN, Wünsch-Filho V. Resolutividade da campanha de prevenção e diagnóstico precoce do câncer bucal em São Paulo, Brasil. Rev Panam Salud Pública. 2007;21(1):30-6. DOI: http://dx.doi.org/10.1590/S1020-49892007000100004
15. Franco EL, Kowalski LP, Oliveira BV, Curado MP, Pereira RN, Silva ME, et al. Risk factors for oral cancer in Brazil: a case-control study. Int J Cancer. 1989;43(6):992-1000. PMID: 2732011 DOI: http://dx.doi.org/10.1002/ijc.2910430607
16. Noce CW, Rebelo MS. Avaliação da relação entre tamanho do tumor e características sociais em pacientes com carcinoma de células escamosas bucal. Rev Bras Cancerol. 2008;54(2):123-9.
17. Jitomirski F. Câncer bucal. In: Pinto VG, org. Saúde bucal coletiva. 4ª ed. São Paulo: Editora Santos; 2000. p.445-56.
18. Koivunen P, Rantala N, Hyrynkangas K, Jokinen K, Alho OP. The impact of patient and professional diagnostic delays on survival in pharyngeal cancer. Cancer. 2001;92(11):2885- 91. PMID: 11753962 DOI: http://dx.doi.org/10.1002/1097-0142(20011201)92:11<2885::AID-CNCR10119>3.0.CO;2-G
19. Morelatto RA, Herrera MC, Fernández EN, Corball AG, López de Blanc SA. Diagnostic delay of oral squamous cell carcinoma in two diagnosis centers in Córdoba Argentina. J Oral Pathol Med. 2007;36:405-8. PMID: 17617833 DOI: http://dx.doi.org/10.1111/j.1600-0714.2007.00547.X
20. Teixeira CM. O consumo de bebidas alcoólicas como lazer [Tese de doutorado]. Vila Real: Universidade de Trás-os-Montes Alto Douro; 2008.
21. Ramos C, Carvalho JEC, Mangiacavalli MASC. Impacto e (i) mobilização: um estudo sobre campanhas de prevenção ao câncer. Ciênc Saúde Coletiva. 2007;12(5):1387-96. DOI: http://dx.doi.org/10.1590/S1413-81232007000500036
22. Sankaranarayanan R, Masuyer E, Swaminathan R, Ferlay J, Whelan S. Head and neck cancer: a global perspective on epidemiology and prognosis. Anticancer Res. 1998;18(6B):4779-86. PMID: 9891557
23. Sciubba JJ. Improving detection of precancerous and cancerous oral lesions. Computer-assisted analysis of the oral brush biopsy. U.S. Collaborative OralCDx Study Group. J Am Dent Assoc. 1999;130(10):1445-57.
24. Myers EN. Otorrinolaringologia cirúrgica - Cirurgia de cabeça e pescoço. 2a ed. Rio de Janeiro: Dilivros; 2011. p.305-32.
25. Franceschi S, Bidoli E, Herrero R, Muñoz N. Comparison of cancers of the oral cavity and pharynx worldwide: etiological clues. Oral Oncol. 2000;36(1):106-15. DOI: http://dx.doi.org/10.1016/S1368-8375(99)00070-6
26. Dhooge IJ, Albers FW, van Cauwenberge PB. Clinical characteristics and diagnostic delay of head and neck cancer: results from a prospective study in Belgium. Eur J Surg Oncol. 1996;22(4):354-8. PMID: 8783651 DOI: http://dx.doi.org/10.1016/S0748-7983(96)90220-6
27. Fritz A, Percy C, Jack A, Shanmugaratnam K, Sobin LH, Parkin MD, eds. International classification of diseases for oncology (ICD-O). 3rd ed. Geneva: World Health Organization; 2000.
28. Sobin LH, Wittekind Ch. Classification of Malignant Tumours. 6ª ed. New Jersey: John Wiley & Sons; 2004. p. 1-44.
29. Detsky AS, McLaughlin JR, Baker JP, Johnston N, Whittaker S, Mendelson RA, et al. What is subjective global assessment of nutritional status? JPEN J Parenter Enteral Nutr. 1987;11(1):8-13. PMID: 3820522 DOI: http://dx.doi.org/10.1177/014860718701100108
30. Detsky AS, Smalley PS, Chang J. The rational clinical examination. Is this patient malnourished? JAMA. 1994;271(1):54-8. PMID: 8258889 DOI: http://dx.doi.org/10.1001/jama.1994.03510250070038
31. Ecog/WHO. Eastern Cooperative Oncology Group/World Health Organization. WHO; 2008.
32. Hashibe M, Brennan P, Benhamou S, Castellsague X, Chen C, Curado MP, et al. Alcohol drinking in never users of tobacco, cigarette smoking in never drinkers, and the risk of head and neck cancer: pooled analysis in the International Head and Neck Cancer Epidemiology Consortium. J Natl Cancer Inst. 2007;99(10):777-89. PMID: 17505073 DOI: http://dx.doi.org/10.1093/jnci/djk179
33. Wünsch-Filho V. The epidemiology of oral and pharynx cancer in Brazil. Oral Oncol. 2002;38(8):737-46. PMID: 12570051 DOI: http://dx.doi.org/10.1016/S1368-8375(02)00066-0
34. Abdo EN, Garrocho AA, Aguiar MCF. Perfil do paciente portador de carcinoma epidermóide da cavidade bucal, em tratamento no Hospital Mário Penna em Belo Horizonte. Rev Bras Cancerol. 2002;48(3):357-62.
35. Gouvea AS, Nogueira MX, Oliveira ZFL, Podestá JRV, Zeidler SVV. Aspectos clínicos e epidemiológicos do câncer bucal em um hospital oncológico: predomínio de doença localmente avançada. Rev Bras Cir Cabeça Pescoço. 2010;39(4):261-5.
36. Farah CS, Ford PJ, Allen K, Vu A, McCullough MJ. Oral Cancer and Potentially Cancerous Lesions - Early Detection and Diagnosis, Oral Cancer. [Acesso em: 2012] Disponível em: http://www.intechopen.com/books/oral-cancer/oral-cancer-and-potentiallycancerous-lesions-early-detection-and-diagnosis
37. IARC - International Agency for Research on Cancer: 2009. Oral cancer/First evidence from a randomized trial that screening reduces oral cancer mortality: The trivandrum oral cancer screening study (TOCS). Kerala, India. [Acesso em: 2011] Disponível em: http://screening.iarc.fr/study_booklptrivandrum.php
38. IARC - International Agency for Research on Cancer: Cervix Cancer Screening. Lyon: IARC Working Group; 2005. p.59-201.
39. Bispo dos Santos AL, Marinho RC, Lima PNM, Fortes RC. Avaliação nutricional subjetiva proposta pelo paciente versus outros métodos de avaliação do estado nutricional em pacientes oncológicos. Rev Bras Nutr Clín. 2012;27(4):243-9.
40. Cuffari L, Tesseroli de Siqueira JT, Nemr K, Rapaport A. Pain complaint as the first symptom of oral cancer: a descriptive study. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;102(1):56-61. PMID: 16831673 DOI: http://dx.doi.org/10.1016/j.tripleo.2005.10.041
41. Abdo EN, Garrocho Ade A, Barbosa AA, Oliveira EL, Franca-Filho L, Negri SL, et al. Time elapsed between the first symptoms, diagnosis and treatment of oral cancer patients in Belo Horizonte, Brazil. Med Oral Patol Oral Cir Bucal. 2007;12(7):469-73.
42. Campos JLG, Chagas JFS, Magna LA. Fatores de atraso no diagnóstico do câncer de cabeça e pescoço e sua relação com sobrevida e qualidade de vida. Rev Bras Cir Cabeça Pescoço. 2007;36(2):65-8.
1. MSc, UTAD, Portugal. (Head and Neck Surgeon, Professor at the UNIMONTES university medical program)
2. PhD. (Full Professor, UTAD - Portugal)
3. MSc and PhD from UNIFESP (Attending and Professor at UNIMONTES)
State University of Montes Claros (UNIMONTES).
Send correspondence to:
Francis Balduino Guimarães Santos
Rua São Pedro, nº 112. Todos os Santos
Montes Claros - MG. Brazil. CEP: 394000-123
Paper submitted to the BJORL-SGP (Publishing Management System - Brazilian Journal of Otorhinolaryngology) on February 16, 2013.
Accepted on July 31, 2013. cod. 10773.