Year: 2001 Vol. 67 Ed. 5 - (13º)
Artigos Originais
Pages: 689 to 693
Prognostic factors on initial glottic cancer
Author(s):
José Carlos de Oliveira 1,
Maria Paula Curado 1,
Abrão Rapoport 2,
Márcio Roberto B. Silva 3
Keywords: glottic cancer, prognosis, survival
Abstract:
Introduction: The survival of initial T1 and T2 glottic cancer after partial surgery or radiotherapy is based on two paradigms: cure and organ preservation. Study design: Retrospective clinical. Material and method: After 72 files revision, 60 (83,3%) men and 12 (16,67%) women, 49 (68,10%) smokers and 32 (49,94%) alcohol consummers, being 30 staged as T1 (41,66%) and 42 as T2 (59,34%), 26 (36,11%) submitted for partial resection (cordectomy, fronto-lateral or hemilaringectomy) and 46 (63,89%) for radiotherapy (medium dosis of 6,3Gy). Results: After a five years followup, the free survival rate for the whole group were 71% (65%) for radiotherapy and 79% for partial resection), with no statistical significance (p=0,4071). For the global survival, partial surgery 91% (12 months), 83% (24 months) and 44% (60 months) for irradiation, 84% (12 months), 68% (24 months) and 17% (60 months), no statistically significant (p=0,0767). Patients with recurrence, underwent total salvage laryngectomy, with 100% survival rate (12 months) and 44% (60 months), similar with results performed after primary resection.
![]()
INTRODUCTION
Laryngeal cancer is the second most common malignant neoplasia of the respiratory tract, second to lung cancer; it is associated with smoking and excessive use of alcohol2, and Tunisia, followed by France and Spain, are the countries that have the highest prevalence rates of this malignant neoplasia14.
As to incidence, there were 18.6/100,000 inhabitants in countries of Asia and Europe14, quite close to the number of cases in São Paulo (17.9/100,000 inhabitants)11. In a study conducted in three Brazilian capitals, the incidence of laryngeal cancer was higher in Porto Alegre when compared to rates in Belém/PA and Goiânia/ GO, justified by the public health policies of the government of Rio Grande do Sul, in which early detection programs were implemented13.
As to glottic cancer, since early symptomatology is evident (especially persistent dysphonia), it is the topographical location responsible for the immediate search for the diagnostic center.
If the disease is diagnosed at its initial stages, survival rates reach 90%, regardless of the type of conservative surgery or radiotherapy, following the paradigm of cleaning the affected area, preserving the laryngeal function (respiration and phonation) and rehabilitating therapeutically the sequelae. It is important to point out that failure in primary treatment may lead to mutilation, since total laryngectomy is the option after initial therapeutic failure in most patients with laryngeal glottic cancer.
The choice of ideal treatment for initial glottic cancer (T1 and T2) depends on clinical variables (staging, general status), demographic variables (age, sex, color and profession), bioethical issues (autonomy, fairness, benefits, malignancy and equity), and personal history (alcohol abuse, smoking), which determine the therapeutic choice. Therefore, in the Head and Neck Surgery at Hospital Araújo Jorge, in Goiânia/ GO, 72 patients with glottic epidermoid carcinoma (Tl and T2) were diagnosed and treated from 1982 to 1996 and the two therapeutic options (partial surgery and radiotherapy) were both considered, as an attempt to define an individual therapeutic protocol for initial glottic cancer, through disease free survival and efficacy of surgical salvage after conservative treatment.
MATERIAL AND METHOD
In the Hospital Araújo Jorge, from Associação de Combate ao Câncer de Goiás/ GO, from 1982 to 1996, we analyzed 72 medical files of initial glottic cancer (T1 and T2), divided into 60 men (83.3%) and 12 women (16.7%), mean age of 59.7 years. As to clinical staging, 30 (41.7%) were T1 and 42 (58.3%) were T2, and 26 (36.1%) were submitted to partial laryngectomy and 46 (63.9%) to radiotherapy (cobalt therapy and linear accelerator at the mean dose of 6.3Gy). Smoking was detected in 49 patients (68.1%), whereas in 8 cases (11.1%) there was no correlation with smoking, and in 15 (20.8%) we did not find any information. As to alcohol abuse, we found it in 32 cases (44.4%), it was denied in 18 (25.1%) and there was lack of information in 22 (30.5%). As to histopathology, the samples consisted of epidermoid carcinomas and in 29 cases (40.3%) it was grade I, in 26 (36.1%) it was grade II and in 13 (18.0%) it was not specified. Data from clinical study were described according to topography and extension of disease.
As to pre-therapeutic routine (oroscopy, anterior and posterior rhinoscopy, indirect and direct laryngoscopy, neck palpation), it also included planigraphy up to 1990 and, after that, laryngeal CT scans were used to complement the TNM classification (1997).
As to radiation therapy, we used daily doses of 180 and 200 cGy for five days per week, with cobalt 60 up to 1980 and linear accelerator of 4 Meve particles after 1985. For the surgical treatment, it included cordectomy, hemilaryngectomy and frontal-lateral laryngectomy.
For statistical planning, we considered the variables sex, age, staging, alcohol abuse, smoking, recurrence, disease free survival and overall survival. In order to correlate treatment and sex, smoking and alcohol abuse, we used chi-square test, with Yates correction. Finally, to analyze overall survival, we employed the statistical method of Kaplan Meyer10.
RESULTS
1. Age, sex, habits and treatment
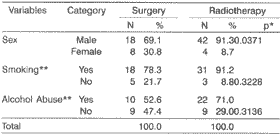
The age of patients submitted to surgery varied from 15 to 71 years (mean age of 57.1 years) and for radiotherapy, it varied from 33 to 82 years (mean age of 61.3 years) - no statistically significant difference (p=0.1702). As to the association sex and therapy, it was relevant (p=0.0371) when we obtained rates of 69.2% and 91.3%, for male patients, respectively for radiotherapy and surgery (partial or salvage). Association of habits (alcohol - p=0.3136, smoking p=0.3228) were not significant (Table 1).
II. Disease free survival; habits and treatment
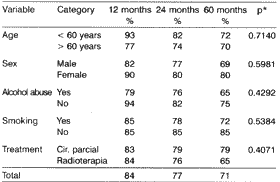
Disease free survival varied from one to 119 months and its variation without recurrence was up to 60 months, with no significance for age (p=0.7140), sex (p=0.5981), alcohol abuse (p=0.4292), smoking (p=0.5384) and therapy used (p=0.4071)(Table 2).
III. Overall survival, habits, demographics and treatment
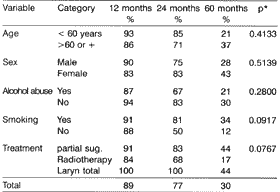
The study of overall survival (Table 3) was made after 12, 24 and 60 months of therapy, observing that up to death there was no statistically significant difference relating to age (p=0.4133), sex (p=0.5139), alcohol abuse (p=0.2800) and smoking (p=0.0917). However, as to partial surgery x radiotherapy, the difference was marginally significant (0.0767). In the partial surgery, survival was 91% (12 months), 83% (24 months) and 44% (60 months), whereas for the radiated group, it was 84% (12 months), 68% (24 months) and 17% (60 months). As to recurrence, overall survival was 100% (12 months), 100% (24 months) and 44% (60 months), similarly to what had happened in the previous group concerning partial surgery.Table 1. Number and percentage of Ti and T2 glottic cancer patients.
* p= descriptive level by chi-square test. ** excluded patients with information ignored.
Table 2. Accumulated properties of disease free survival in T1 and T2 glottic cancer patients.
* p= descriptive level of log-rank test
Table 3. Probability of overall survival accumulated in patients submitted to radiotherapy and partial surgery for T1 and T2 vocal fold cancer.
DISCUSSION
The analysis of the sample of initial glottic cancer (T1 and T2) patients enabled some confirmations if we considered the variables individually or associated, determining overall survival and disease free survival of the 72 analyzed patients.
As to age, glottic cancer patients are normally in the 6th decade of life and they have co-morbidity, such as hypertension, diabetes and chronic pulmonary disease, which increases the risk of postoperative complications. In our study, the mean age was 59.7 years, but the literature proposed 62.5 years15,17,22.
Prevalence in male patients was evident (5:1), and it may be explained by smoking, in accordance with the literature15,17,22. The predominance of male subjects (60 men to 12 women) was significant (p=0.0371), and 49 of them were smokers and 8 were not, whereas in 15 subjects, there were no references (p=0.3228) clearly showing the association between gender and smoking in glottic cancer.
As to co-carcinogenic agents, Table 1 shows that 71% of the patients abused of alcohol, which was not statistically significant (p=0.3130), but it is normally associated with smoking and it becomes a co-factor providing a 43-fold increase of glottic cancer1. As far as smoking goes, 91.2% of the cases were smokers and it is known that the relative risk of glottic cancer is 10 times higher in smokers19.
In the prognostic evaluation, disease free survival varied from one to 119 months, and for up to 60-month recurrence (Table 2) there were no statistically significant differences concerning age (p=0.7140), alcohol abuse (0.4292), smoking (p=0.5384) and treatment (p=0.4071). Patients submitted to radiotherapy had five-year disease free survival of 65% and 79% for partial surgery, close to the figures provided by the literature (70% and 90%), depending on being TI or T23,5,6,7,8,16,18,20,21.
We believe that differences in disease free survival reported in all series are a result of incorrect staging of tumors, surgical inexperience and radiotherapy planning. In our experience, the absence of difference between disease free survival in surgical and radiotherapy groups should be considered true, since age and type of treatment were not relevant.
Overall survival was equivalent for the surgical group and the radiotherapy group, respectively 89%, 77% and 30%, for 12, 24 and 60 months. However, we detected differences concerning vocal quality, which was clearly better in our experience, similarly to the literature17,19. As to relative risk of death, it was 1.9 times higher in the radiotherapy group; the patients with recurrence salvage surgery did not modify the relative risk of death, as compared to the group submitted to partial surgery.
Based on everything that was analyzed in the present retrospective study, a number of critical considerations were made:
1. The length of anterior commissure (non-existent in T1a and T2b) patients, with or without invasion of supra and subglottis, was not always considered in the adopted clinical staging (TNM).
2. Currently, in the length of anterior commissure (T1b), indication for partial surgery is preferred; however, if it is not performed (T1a), radiotherapy becomes the preferred choice.
3. If there is supra or subglottis invasion, in the presence of vocal fold mobility (T2b), surgery is the preferred treatment because it provides better cure chances and lower incidence of recurrence.
As to surgical salvage, in our sample, there was five-year survival in 44% of the patients submitted to total laryngectomy post-partial initial resection and 17% after initial radiation. It is important to point out that surgical salvage does not compromise the indication of partial surgery, and survival is 100% after 24 and 60 months.
Another relevant consideration is indication of partial salvage surgery after radiotherapy. The literature brings figures from 70 to 80% of 5 and 10-year survivals after fronto-lateral and laryngectomy12, and 100% after crico-hyoidpexia9.
Therefore, we can consider surgical salvage as a new paradigm for the treatment of irradiated and relapsed glottic cancer, enabling a conservative strategy.
CONCLUSIONS
To sum up, for T1a tumors, irradiation and, more recently, laser microsurgical resections tend to predominate4. For T1b tumors, in which the commissure is affected, partial laryngectomy is preferred. For T2a tumors, radiation is indicated as a routine, and for T2b patients, partial laryngeal surgeries are advocated. Nevertheless, overall and disease free survivals are marginally similar in the treatment of initial laryngeal cancer, regardless of the therapy.
REFERENCES
1. BERRINO, F.; CROSIGNANI, P. - Epidemiology of Malignant Tumors of the Larynx and Lung. Ann Ist Super Sanitá, 28:107-20, 1992.
2. CATARRUZA, M.S.; MAISSONNEUVE, P.; BOYLE, P. - Epidemiology of Laryngeal Cancer Oral Oncology. Eur J Cancer, 32b:293-305, 1996.
3. CHEN, W.C.; CHEN, L.H.; LEUNG, W.M.; TANG, S.G.; TSAI, M.H.; LIAO, C.T.; SEE, L.C. - Comparison of radiotherapy and surgical results of early stage glottic cancer: a retrospective analysis. J Formos Med Assoc, 92:1076-83, 1993.
4. DINARDO, L.J.; KAYLE, D.M.; ISAACSON, J. - Currents treatment practices for early laryngeal carcinoma. Otolaryngol Head Neck Surg, 12030-7, 1999.
5. HOGIKYAN, N.D.; BASTIAN, R.W. - Surgical therapy of glottic and subglottic tumors. In LINDENBERG, R.D.; THAWLEY, S.E.; BATSAKIS, J.G.; PANJE, W.R. - Comprehensive management of head and neck tumors. 2 ed., Philadelphia, PA, W B Sauders Co., 2:1039 68, 1999.
6. JOHANSEN, L.V.; OVERGAARD, J.; HJELM-HANSEN, M.; GADBERG, C.C. - Primary radiotherapy of T1 squamous cell carcinoma of the larynx: analysis of 478 patients treated from 1963 to 1985. Int J Radiat Oncl Biol Phys, 18:1307-13, 1990.
7. KELLY, M.D.; SPAULDING, C.A.; CONSTABLE, W.C.; HAHN, S.S.; KERSH, C.R.; CANTRELL, R.W. - Definitive radiotherapy in the management of stage I and II carcinomas of the glottis. Am Otol Rhinol Laryngol, 98:235-9, 1989.
8. KOWALSKI, L.P.; BATISTA, M.B.P.; SANTOS, C.R.; SCOPELLA, A.; SALVAJOLI, J.V.; NOVAES, P.E.; TRIPPE, N. - Prognostic factors in glottic carcinoma clinical stage I and II treated by surgery or radiotherapy. Amer J Otolaryngol, 14:122-7, 1993.
9. LACCOURREYE, H. - La chirurgie partielle des epitheliomas de l'étage glottique. Evolution de nos indications. Bull Acad Natle Méd, 177:1333-41, 1993.
10. LEE, E.T. - Statistical methods for survival data analysis. 2nd ed. New York: John Wiley & Sons INC, 63-218, 1992.
11. MIRRA, A.P.; FRANCO, E.L. - Incidência de Câncer no Município de São Paulo Brasil. São Paulo: Instituto Ludwig de Pesquisas Contra o Câncer, 119, 1985.
12. NIBU K.; KAMATA, S.; KAWABATA, K.; NAKAMIZO, M.; NIGAURI, T.; HORI, K. - Partial laryngectomy in the treatment of radiationfailure of early glottic carcinoma. Head & Neck, 19:116-20, 1997.
13. PARKIN, D.M.; WHELAN, S.L.; FERLAY, J; RAYMOND, L; YOUNG, J. - Cancer Incidence in Five Continents. v.VII IARC Scientific Publications n. 143, Lyon. 1240, 1997.
14. PISANI, P.; PARKIN, D.M.F.; FERLAY, J. - Estimates of the worldwide mortality of 25 cancer. Int J. of Cancer, 83:18-29, 1990.
15. RIES, L.A.G.; HANKEY, B.F.; HARRAS, A.; DEVESA, S.S. - Cancer incidence, mortality and patient survival in the United States. In SCHOTTENFELD, D.; FRAUMENI, J.F. Jr. - Cancer Epidemiologyand Prevention. Second edition Oxford University Press, 1996, 168 91.
16. RUCCI, L.; GALLO, O.; FINI-STORCHI, O. - Glottic cancer involving anterior commissure: surgery vs. radiotherapy. Head & Neck, 13:403 10, 1991.
17. SESSION, R.B.; HARRISON, L.B.; O'MALLEY, B.B. - Early cancer of the Larynx. In HARRISON, L.B.; SESSIONS, R.B.; HONG, WK. - Head and neck cancer a multidisciplinary approach. Philadelphia, PA.: Lippincott-Raven Publishers, 481-509, 1999.
18. SMEE, R.; BRIDGER G.P.; WILLIAMS, J.; FISHER, R. - Early glottic carcinoma: results of treatment by radiotherapy. Australas Radiol, 44:53-59, 2000.
19. TUNYS, A. J. et al. - Cancer of the larynx/hypopharynx, tobacco, and alcohol. IARC International Case control Study in Turin and Varese (Italy), Zaragoza and Navarra (Spain), Geneva (Switzerland) and Calvados (France). Int J Cancer, 41:483-91, 1988.
20. TURESSON, L; SANDENBERG, N.; MERCKE, K-A, JOHANSSON,; SANDIN, L; WALGREN, A. - Primary radiotherapy for glottic laryngeal carcinoma stage I and II. Acta Oncol, (Sweden) 30:357-62, 1991.
21. VEGA, S.F.; SCOLA, B.; VEGA, M.F.; MARTNEZ, T.; SCOLA, E. Chirurgia verticalle delta laringe. Tecnica chirurgica. Risultati oncologici a funcionali. Acta Otorhinol., 16272-80, 1996.
22. WATERHOUSE, J.A.H. - Epidemiolgy. In FERLITO, A. Neoplasms of the Larynx. London United Kingdom, Churchill Livingstone, 49-64, 1993.
1 Master in Head and Neck Surgery, Hospital Heliópolis, Hosphel, São Paulo/ SP.
2 Full Professor, Head and Neck Surgery, Hospital Heliópolis, Hosphel, São Paulo/ SP.
3 Surgeon, Head and Neck Surgery, Hospital Araújo Jorge, Goiânia/ GO.
Study conducted at Hospital Araújo Jorge, Goiânia/ GO.
Address correspondence to: Prof. Dr. Abrão Rapoport - Praia Amadeu Amaral, 47 - cjt°. 82 - Paraiso - 01327-010 São Paulo/ SP - Brazil.
E-mall: cpgcp.hosphel@attglobal.net
Article submitted on March 28, 2001. Article accepted on May 30, 2001.