Year: 2014 Vol. 80 Ed. 2 - (5º)
Artigo Original
Pages: 105 to 110
Postural control in underachieving students
Author(s): Andreza Tomaz1; Maurício Malavasi Ganança2; Adriana Pontin Garcia1; Natalia Kessler1; Heloisa Helena Caovilla3
DOI: 10.5935/1808-8694.20140024
Keywords: Postural balance; Underachievement; Vestibular function tests
Abstract:
Postural balance is a sensory-motor function resulting from a learning process.
OBJECTIVE: To evaluate the postural control of underachieving students through static posturography together with virtual reality stimulation.
METHODS: This was a controlled cross-sectional study of a group of 51 underachieving students and a control group of 60 students with good school performance, with no history of vestibular disorders or neurotological complaints, volunteers from the community, age- and gender-matched. The students were submitted to Balance Rehabilitation Unit (BRUTM) posturography.
RESULTS: A total of 111 students aged 7 to 12 years old were evaluated. At posturography evaluation, there was no significant difference between the limit of stability area (cm2) of the control group and the experimental group. The comparison between groups demonstrated a statistically significant difference (p < 0.05) in the values of sway velocity (cm/s) and center of pressure area (cm2) in the ten sensory conditions evaluated.
CONCLUSION: Posturography with virtual reality stimulation, allows for the identification of incapacity to maintain postural control, with or without visual deprivation, and the assessment of visual, somatosensory, and vestibular-visual interaction conflict in underachieving students.
![]()
INTRODUCTION
Postural balance is a sensory-motor function that depends on information from the vestibular, visual, and somatosensory systems. The central nervous system integrates these signals, generating eye movements coordinated with head movements to maintain a clear image; performs adjustments on the position and movements of the head and body in relation to gravity, to the support surface and to the environment in order to maintain upright posture; and helps in static and dynamic spatial orientation, improving motor performance and locomotion.1
In children, at approximately the age of 7 years, the postural control system ceases to be strictly dependent on vision and becomes part of the information originating from the vestibular and somatosensory system, assuming a similar strategy to that of adults.2
Motor activity is very important for the overall development of the child, since self-awareness and the perception of body, space, and time result from exploring the surrounding world, which represent the basic components for learning and development of school-related activities.3
Dizziness, identified in 15.0% of school-age children,4 and other types of dizziness, imbalance, falls, neurovegetative manifestations, visual distortion, headache, excessive tiredness, restlessness, sleep disturbance, sleep disorders, motion sickness, sometimes associated with hearing loss, tinnitus, and feeling of pressure in the ear can affect children and adolescents5-6 and interfere with psychological behavior and school performance.7-9
Vestibular disorders may impair motor development in school-age children, preventing appropriate contact with the environment and the acquisition and development of language, affecting learning and communication skills, and hindering the ability to speak, read, write, spell, and perform mathematical calculations.9-12
Children with vestibular dysfunction may have difficulty performing physical exercises, have distorted perceptions of body size and surrounding objects, fail to realize the extent of their limbs, and may be inattentive. The learning disorder can occur due to the incapacity to perform coordinated movements and inaccurate conception of spatial position.13
The maintenance of postural stability in children with vestibular disorders may be more difficult while reading, a complex task that requires several processes, such as perception, eye movement, and linguistic and semantic skills.14
The diagnosis of vestibular disorders in childhood is a challenging task, as children cannot describe their symptoms accurately.10,15-17
With suspicion of a vestibular impairment, the child should be submitted to an otoneurological assessment to confirm alterations and to determine the impaired side and the intensity of the problem; these data can help in therapeutic decision-making.18
Electronystagmography (ENG), vectoelectronystagmography (VENG), or videonystagmography (VNG) analyze the vestibular system and the neural structures involved in maintaining body balance through different vestibular and oculomotor tests.18-19 Relevant vestibular alterations have been observed in children with learning difficulty complaints.6,20-22
Computerized posturography complements the otoneurological diagnostic assessment, allowing for the identification of abnormal signals in patients with complaints of balance disorders. It can be performed on static (stabilometry or statiokinesiometry) and dynamic force platforms (dynamic posturography); its value in the study of vestibular-spinal reflex (VSR) is similar to the use of electronystagmography to study the vestibular-ocular reflex (VOR).23,24
Computerized posturography includes vestibular, visual, and somatosensory information in order to: detect changes in postural balance in individuals of all ages;17 improve the diagnostic rate of patients with complex balance disorders, identify systems involved earlier with greater sensitivity and specificity, and document and monitor the treatment of balance disorders.25
Posturography can add important quantitative data on the position of the center of pressure relative to the base of support in different sensory conditions for the vestibular assessment of children and adolescents with dizziness.17,26
Posturography with virtual reality is a new method; it provides visual stimuli that trigger oculomotor pursuit, saccadic, optokinetic, and vestibular-visual interaction responses.27-31 Virtual reality allows for an immersion into an illusory world, where the perception of the environment is modified by an artificial sensory stimulus that can cause ocular-vestibular conflict and modify the VOR gain.32 Posturography integrated with virtual reality may be useful in differential diagnosis and therapeutic interventions in patients complaining of dizziness.33 The posturography module of the Balance Rehabilitation Unit (BRUTM) projects stimuli in virtual reality goggles and provides information on the position of the patient's center of pressure in ten sensory conditions, through quantitative indicators, the limit of stability area, the center of pressure area, and body sway velocity.34
The interest in assessing the parameters of static posturography with virtual reality stimuli in underachieving students arose from the fact that a literature search retrieved no pertinent articles on this subject, and the possibility that any abnormal findings might have diagnostic and therapeutic implications.
This study aimed to evaluate the postural control of underachieving students through static posturography integrated with virtual reality.
METHOD
This contemporary cross-sectional cohort study was performed after approval by the institution's research ethics committee, under protocol number 1635/09. All students and guardians received information about the content of the research through an explanatory letter and signed an informed consent before the investigation was started.
The experimental group consisted of male and female underachieving students from public and private elementary schools, aged between 7 and 12 years, attended to at the Outpatient Clinic of Language of the Department of Human Communication Disorders; the control group consisted of volunteer students from the community, from public and private elementary schools, with good school performance and no history of vestibular disorders or neurotological complaints, who were age- and gender-matched.
The criterion for inclusion of students in the experimental group was underachievement without abnormalities on tonal and vocal audiometry. Underachievement was considered when the student had insufficient grades in different subjects of the school curriculum, lower than the expected for age, schooling, and cognitive skills, according to parents', teachers', or school coordinators' reports. The criterion for inclusion of students in the control group was good academic performance, absence of symptoms related to body balance and/or hearing, and no abnormalities on tonal and vocal audiometry. Good academic performance was considered when the student earned good grades in school subjects, presenting the expected school performance for age, cognitive skills, and schooling, according to parents', teachers', or school coordinators' reports.
In both groups, the exclusion criteria were students unable to understand and answer simple verbal commands; those unable to stand independently in the upright position; those with severe visual impairment or not corrected by the use of lenses; orthopedic disorders that resulted in limited movement or prostheses in the lower limbs; those with psychiatric disorders, using drugs acting on the central nervous system or the vestibular system; those undergoing rehabilitation of postural balance in the last six months; and those with headache complaints.
Students underwent a neurotological assessment consisting of anamnesis, which investigated the occurrence of dizziness and other neurotological symptoms; visual inspection of the external auditory meatus; tonal and vocal audiometry (Maico audiometer, Model MA41); functional assessment of the vestibular system; and BRUTM posturography.
To undergo the vestibular system functional test, all students were instructed to abstain from coffee, tea, and chocolate; to avoid using any non-essential medication that could interfere with results for 72 hours before the examination; and to abstain from eating or drinking for three hours before the test. The equipment used was a VECWIN for VENG and a Neurograff Eletromedicina Ind. e Com. - EPT - Brazil air otocalorimeter (24º and 50ºC). The VENG included eye movement calibration; spontaneous and semi-spontaneous, positional and positioning nystagmus test; saccadic movements; pendular tracking; optokinetic nystagmus; pendular rotary chair; and caloric test.18,19,35,36
BRUTM posturography included a computer with assessment program, a metallic safety structure, a protective pad with handles and seatbelt, a force platform, virtual reality goggles with light protection, an accelerometer, and a foam pad. The balance platform converted the pressure applied on the surface into electrical signals to determine the position of the center of pressure through quantitative indicators: the area of the limits of stability and center of pressure area, and sway velocity in ten sensory conditions. The center of pressure area 95% confidence interval was defined as the area of distribution of 95% of the samples from the center of pressure. The mean sway velocity was determined by the total distance divided by the 60 seconds of test duration.
To determine the limit of stability, students were instructed to perform maximum body displacements in the anteroposterior and latero-lateral directions through the ankle strategy, without moving the feet and without using trunk and/or hip strategies. Instructions were given to the students to move slowly, as much as possible without losing balance, in the following sequence: a) forward; b) return to starting position; c) to the right; d) return to starting position; e) to the left; f) return to starting position; g) backward; and h) return to starting position. The students were asked to perform this sequence of movements twice, without necessarily using the 60 seconds reserved for this procedure. The procedure was repeated when the foot was moved or when trunk and/or hip movements were used.27-31,34
To determine the center of pressure area and body sway velocity, students were instructed to remain in standing position without moving the upper limbs, ankles, and/or feet, with arms extended along the body for 60 seconds in each of the ten sensory conditions: 1) on solid surface, with eyes open; 2) on solid surface, with eyes closed; 3) on foam surface, with eyes closed; 4) on solid surface, with saccadic stimulation; 5) on solid surface, with optokinetic stimulation, in the horizontal direction (left to right); 6) on solid surface, with optokinetic stimulation, in the horizontal direction (right to left); 7) on solid surface, with optokinetic stimulation, in the vertical direction (top to bottom); 8) on solid surface, with optokinetic stimulation, in the vertical direction (bottom-up); 9) on solid surface, with optokinetic stimulation, in the horizontal direction, associated with slow and uniform head rotation movements; 10) on solid surface, with optokinetic stimulation, in the vertical direction associated with slow and uniform flexion-extension head movements.29 Resting intervals were provided according to students' necessity; safety was ensured by the presence of the examiner next to the student throughout the evaluation in order to avoid risks, such as possible fall.
A descriptive statistical analysis was performed for sample characterization. The chi-squared test was used to analyze the homogeneity of gender proportions between the control and experimental groups. Levene's test was used to analyze the equality of variances regarding age. Student's t-test was used to compare the experimental and control groups regarding age. The Mann-Whitney test was used to compare the control and experimental groups on the limits of stability, sway velocity, and the center of pressure area in the ten conditions of static posturography of the BRUTM, as the assumption of normal distribution was rejected the Shapiro-Wilk's test. Data were shown as mean ± standard deviations, medians, and minimum and maximum values. The level of significance was set at p < 0.05. The Predictive Analytics Software (PASW, release 18.0) was used for calculations.
RESULTS
A total of 111 students were assessed. The control group consisted of 36 male and 24 female students, mean age of 9.6 ± 1.6 years. The experimental group consisted of 36 male and 15 female students, mean age of 9.3 ± 1.5. There was no significant difference between groups regarding gender (p = 0.335) and age (p = 0.056).
Dizziness or other neurotological symptoms were not reported by the students in either group.
The posturography assessment showed no significant difference (p = 0.894) between the values of the limits of stability (cm2) in the control group (mean ± SD = 195.7 ± 66.9; median = 182.5; minimum-maximum value = 72.0 to 399.0) and the values in the experimental group (mean ± SD = 194.5 ± 75.2; median = 196.0; minimum-maximum value = 65.0 to 389.0).
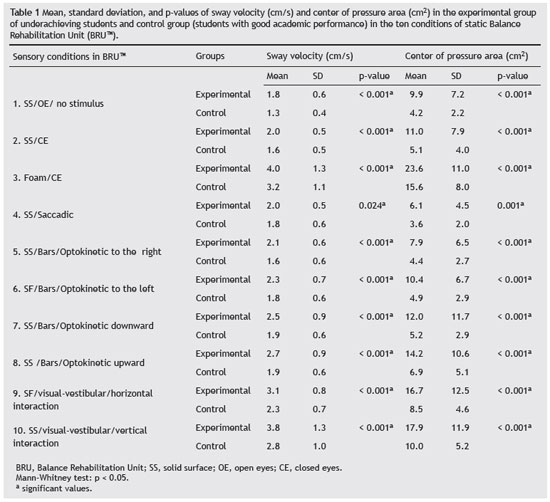
Table 1 presents the mean, standard deviation, and p-value of the sway velocity (cm/s) and the center of pressure area (cm2) in the ten conditions of static BRUTM posturography according to the group. The mean values of sway velocity and the center of pressure area in the experimental group were significantly higher than those in the control group in the ten sensory conditions evaluated.
DISCUSSION
Static posturography, combined with BRUTM virtual reality, was used in this study to evaluate the postural control of a group of underachieving students compared with a group of students with good academic performance and no otoneurological complaints. The groups were homogeneous regarding gender and age. Students from both groups reported no dizziness or other neurotological symptoms. All students underwent the procedure properly and without difficulty.
In the present series, static posturography demonstrated that the values of the limits of stability in the experimental group area were similar to those in the control group. Therefore, the group of underachieving students had the capacity to adequately control the maximum center of pressure sway on the platform. We could not find any references in the literature that discussed the limits of stability for underachieving students on static posturography combined with BRUTM.
The increased values of the center of pressure area and sway velocity in asymptomatic underachieving students suggest incapacity to maintain postural control with or without deprivation of sight and in visual, somatosensory, and visual-vestibular interaction conflict. Signs of vestibular dysfunction without neurotological symptoms also have been observed in some children with language disorders or15,37 underachievement.11
Underachievement suggests involvement of the vestibular system, which must necessarily be excluded. Many children with vestibular dysfunction do not have any symptoms related to body balance, which could be explained by concomitant autocompensation in slowly progressive lesions, and would justify the absence of vestibular symptoms, frequently observed in chronic vestibular disorders or difficulty to understand a description, express what they feel, or remember what they felt in the past.6-7,15,37
The values of the center of pressure area and sway velocity for the static BRUTM posturography in the group with underachievement were higher than those of the control group in all ten assessed sensory situations. Therefore, the static BRUTM posturography was able to capture the incapacity to maintain postural control in students without symptoms of change in body balance. We were unable to find any literature references on changes in values of the center of pressure area and sway velocity for underachieving students in static BRUTM posturography.
The present findings for underachieving students in static BRUTM posturography do not allow for adequate quantitative comparisons with the results of other types of posturography, since the methods and evaluated parameters are different.
The literature search demonstrated that there is little information on the use of posturography in children, and no studies on underachieving students were retrieved.
However, some authors have studied the postural balance of normal children using other types of posturography with the addition of dual-task conditions,38,39 vibration of the ankle tendons as additional somatosensory stimulation,40 or an oscillator attached to a platform on the ground.41
In studies of dyslexic children compared with non-dyslexic, static posturography with the Techno Concept® demonstrated greater postural instability and increased center of pressure area, with or without visual, cognitive, or proprioceptive activities.14,42-45
The present findings demonstrate that posturography integrated to virtual reality provides relevant data on postural control in underachieving students. The characterization of body balance disorder in these students has diagnostic, therapeutic, and even preventive implications.
Further studies should be conducted in this area in order to better understand the association between postural control and underachievement.
CONCLUSION
Posturography, together with virtual reality, allows for the identification of signs of incapacity to maintain postural control, with or without visual deprivation, in visual, somatosensory, and vestibular-visual interaction conflict in underachieving students.
FUNDING
This study was supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - CAPES.
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
REFERENCES
1. Caovilla HH, Ganança MM. Princípios e indicações da reabilitação vestibular. In: Costa SS, Tsuji DH, Lessa MM, Cruz OLM. Pro-ORL/ABORL-CCF, módulo 4. Porto Alegre: Artmed/Panamericana; 2010. p. 23-61.
2. Woollacott M, Debu B, Mowatt M. Neuromuscular control of posture in the infant and child: is vision dominant? J Mot Behav. 1987;19:167-86.
3. Rosa Neto F. Manual de avaliação motora. Porto Alegre: Artmed; 2002. p.11-27.
4. Russel G, Abu-Arafeh I.Paroxysmal vertigo in children - an epidemiological study. Int J Pediatr Otorhinolaryngol. 1999;49:S105-7.
5. Deus LHR, Ganança CF, Ganança FF, Ganança MM, Caovilla HH. Sintomas otoneurológicos em crianças e adolescentes com distúrbios de linguagem. Acta ORL. 2008;26:118-23.
6. Ganança MM, Caovilla HH, Munhoz MSL, Ganança FF. Tonturas na criança e no adolescente. Rev Bras Med Otorrinolaringol. 1995;2:217-42.
7. Ganança MM, Caovilla HH. Correlação entre distúrbio de linguagem e disfunção labiríntica. Pediatr Modern. 1990;25:128-45.
8. Campos MI, Ganança FF, Caovilla HH, Ganança MM. Prevalência de sinais de disfunção vestibular em crianças com vertigem e/ou outros tipos de tontura. Braz J Otorhinolaryngol. 1996;3:165-71.
9. Formigoni LG, Medeiros IRT, Santoro PP, Bittar RSM, Bottino MA. Avaliação clínica das vestibulopatias na infância. Braz J Otorhinolaryngol. 1999;65:78-82.
10. Ganança MM, Caovilla HH, Munhoz MSL, Munhoz ML, Ganança FF. Tratamento da vertigem na criança. Pediatr Modern. 1997;33:7-22.
11. Ganança CF, Pupo AC, Caovilla HH, Ganança MM. Disfunção vestibular em crianças e adolescentes com mau rendimento escolar. Fono Atual. 2000a;11:21-7.
12. Franco ES, Panhoca I. Sintomas vestibulares em crianças com queixa de dificuldades escolares. Rev Soc Bras Fonoaudiol. 2008;13:362-8.
13. McHugh HE. Auditory and vestibular disorders um children. Laryngoscope. 1962;72:555-65.
14. Legrand A, Bui-Quoc E, Doré-Mazars K, Lemoine C, Gérard CL, Bucci MP. Effect of a dual task on postural control in dyslexic children. PLoS One. 2012;7:e35301.
15. Ganança MM. Da vestibulometria em crianças com distúrbios de linguagem. Parte 1. Pro Fono. 1989;1:3-11.
16. Ramos MEVC, Azevedo APM, Lengruber A, Campos A, Chiari EP. Roteiro de avaliação otoneurológica infantil do recém-nato até a criança de 12 anos. Braz J Otorhinolaryngol. 1997;4:164-8.
17. Medeiros IRT, Bittar RSM, Pedalini MEB, Lorenzi MC, Kii MA, Formigoni LG. Avaliaçäo do tratamento dos distúrbios vestibulares na criança através da posturografia dinâmica computadorizada: resultados preliminares. J Pediatr. 2003;79:337-42.
18. Ganança MM, Caovilla HH, Ganança FF. Electronystagmography versus videonystagmography. Braz J Otorhinolaryngol. 2010;76:399-403.
19. Caovilla HH, Gananca CF. Avaliação do Equilíbrio Corporal: Conceituação e aplicação clínica. In: Bevilacqua MC, Martinez MAN, Balen AS, Pupo AC, Reis ACM, Frota S. Tratado de audiologia. São Paulo: Santos; 2011. p. 317-29.
20. Zeigelboim BS, Dalcumune I, Manzonl M, Jurklewlcz AL, Martins-Bassetto J, Klagenberg KF. Avaliação vestibular em crianças com distúrbio de aprendizagem. Pediatr Modern. 2006;42:215-2.
21. Franco ES, Panhoca I. Avaliação otoneurológica em crianças com queixas de dificuldades escolares: pesquisa da função vestibular. Braz J Otorhinolaryngol. 2007;73:803-15.
22. Sousa EC, Síller AL, Tuma VC, Ganança CF, Ganança MM, Caovilla HH. Relação entre dificuldade de leitura e escrita e sintomas e sinais de vestibulopatia periférica em crianças em idade escolar. ORL. 2008;26:112-7.
23. Barona R, Garin L, Comeche C. Estudio del reflejo vestibuloespinal. Aplicaciones clinicas de la posturografia. Acta Otorrinolaringol Esp. 1993;44:217-21.
24. Harcourt JP. Posturography: applications and limitations in the management of the dizzy patient. Clin Otolaryngol Allied Sci. 1995;20:299-302.
25. Bittar RSM. Como a posturografia dinâmica computadorizada pode nos ajudar nos casos de tontura? Arq Int Otorrinolaringol. 2007;11:330-3.
26. Hubbell RN, Skoner JM. Vertigo in children. In: Fabian RL, Gluckman JL, Donald P, eds. Current Opinion in Head and Neck Surgery. Philadelphia: Lippincott, Williams & Wilkins; 1999.p.328-34.
27. Gazzola JM, Dona F, Ganança MM, Suarez H, Ganança FF, Caovilla HH. Realidade virtual na avaliação e reabilitação dos distúrbios vestibulares. Acta ORL. 2009;27:22-7.
28. Cusin FS, Ganança MM, Ganança FF, Ganança CF, Caovilla HH. Balance Rehabilitation Unit (BRUTM) posturography in Menière's disease. Braz J Otorhinolaryngol. 2010;76:611-7.
29. Kessler N, Ganança MM, Ganança CF, Ganança FF, Lopes SC, Serra AP, et al. Balance Rehabilitation Unit (BRUTM) posturography in relapsing-remitting multiple sclerosis. Arq Neuropsiquiatr. 2011;69:485-90.
30. Monteiro SR, Ganança MM, Ganança FF, Ganança CF, Caovilla HH. Balance Rehabilitation Unit (BRUTM) posturography in benign paroxysmal positional vertigo. Braz J Otorhinolaryngol. 2012;78:98-104.
31. Moreira DA, Ganança MM, Caovilla HH. Static posturography in addicted to illicit drugs and alcohol. Braz J Otorhinolaryngol. 2012;78:97-103.
32. Di Girolamo S, Picciotti P, Sergi B, Di Nardo W, Paludetti G, Ottaviani F. Vestibulo-ocular reflex modification after virtual environment exposure. Acta Otolaryngol. 2001;121:211-5.
33. Keshner EA, Streepey J, Dhaher Y, Hain T. Pairing virtual reality with dynamic posturography serves to differentiate between patients experiencing visual vertigo. J Neuroeng Rehabil. 2007;4:24.
34. BRUTM. Unidade de Reabilitação do Equilíbrio. Manual do usuário. Versão 1.0.7. Versão do Software: 1.3.5.0. Uruguai: Medicaa; 2006. 132p.
35. Albertino S, Bittar RSM, Bottino MA, Ganança MM, Gonçalves DU, Greters ME, et al. Valores de referência da prova calórica a ar. Braz J Otorhinolaryngol. 2012;78:2.
36. Barros ACMP, Caovilla HH. From nystagmus to the air and water caloric tests. Braz J Otorhinolaryngol. 2012;78:120-5.
37. Ganança MM. Da vestibulometria em crianças com distúrbios de linguagem. Parte 2. Pro Fono. 1990;2:3-8.
38. Blanchard Y, Carey S, Coffey J, Cohen A, Harris T, Michlik S, et al. The influence of concurrent cognitive tasks on postural sway in children. Pediatr Phys Ther. 2005;17:189-93.
39. Schmid M, Conforto S, Lopez L, D'Alessio T. Cognitive load affects postural control in children. Exp Brain Res. 2007;179:375-85.
40. Cuisinier R, Olivier I, Vaugoyeau M, Nougier V, Assaiante C. Reweighting of sensory inputs to control quiet standing in children from 7 to 11 and in adults. PLoS One. 2011;6:e19697.
41. Fujiwara K, Kiyota T, Mammadova A, Yaguchi C. Age-related changes and sex differences in postural control adaptability in children during periodic floor oscillation with eyes closed. J Physiol Anthropol. 2011;30:187-94.
42. Pozzo T, Vernet P, Creuzot-Garcher C, Robichon F, Bron A, Quercia P. Static postural control in children with developmental dyslexia. Neurosci Lett. 2006;403:211-5.
43. Kapoula Z, Bucci MP. Postural control in dyslexic and non-dyslexic children. J Neurol. 2007;254:1174-83.
44. Vieira S, Quercia P, Michel C, Pozzo T, Bonnetblanc F. Cognitive demands impair postural control in developmental dyslexia: a negative effect that can be compensated. Neurosci Lett. 2009;462:125-9.
45. Quercia P, Demougeot L, Dos Santos M, Bonnetblanc F. Integration of proprioceptive signals and attentional capacity during postural control are impaired but subject to improvement in dyslexic children. Exp Brain Res. 2011;209:599-608.
1. Post-Graduate Program in Human Communication Disorders, Universidade Federal de São Paulo (UNIFESP), São Paulo, SP, Brazil
2. Department of Otorhinolaryngology, Head and Neck, Universidade Federal de São Paulo (UNIFESP), São Paulo, SP, Brazil
3. Discipline of Otology and Otoneurology, Universidade Federal de São Paulo (UNIFESP), São Paulo, SP, Brazil
Corresponding author.
A. Tomaz
E-mail: andreza.tomaz@h2oviva.com.br
Received 29 July 2013.
Accepted 17 November 2013.