



Trabalho Clínico
Retalho pediculado de corneto inferior: um novo retalho vascularizado para reconstrução de base de crânio
The posterior pedicle inferior turbinate flap:
a new vascularized flap for skull base reconstruction
Autores:
Felipe Sartor Guimarães Fortes (Ex-Médico Residente e Preceptor do HCFMUSP. Fellowship Universidade de Pittsburgh. ) Pós-Graduando da Disciplina de Otorrinolaringologia da FMUSP>
Ricardo L. Carrau (MD ) Professor of the Department of Otolaryngology and Head & Neck Surgery - University of Pittsburgh (UPMC), Minimally Invasive endoNeurosurgery Center University of Pittsburgh Medical Center
Carl H. Snyderman (MD) Professor of the Department of Otolaryngology and Head & Neck Surgery - University of Pittsburgh (UPMC), Minimally Invasive endoNeurosurgery Center University of Pittsburgh Medical Center
Daniel Prevedello (MD) Fellowship of the Minimally Invasive endoNeurosurgery Center
University of Pittsburgh Medical Center
Allan Vescan (MD) Fellowship of the Minimally Invasive endoNeurosurgery Center University of Pittsburgh Medical Center
Arlan Mintz (MD) Department of Neurological Surgery - UPMC,
Minimally Invasive endoNeurosurgery Center University of Pittsburgh Medical Center
Paul Gardner (MD) Department of Neurological Surgery - UPMC, Minimally Invasive endoNeurosurgery Center University of Pittsburgh Medical Center
Amin B.Kassam (MD) Chairman Department of Neurological Surgery - UPMC, Minimally Invasive endoNeurosurgery Center University of Pittsburgh Medical Center
Carlos Diógenes Pinheiro Neto (Médico Otorrinolaringologista) Médico Preceptor do HC-FMUSP
Palavras-Chave
Cirurgia endoscópica, Base de crânio, Fístula liquórica, Reconstrução, Corneto inferior, Retalho vascularizado
Resumo
Introdução. O uso dos acessos endoscópicos endonasais (EEA) para ressecção de lesões da base do crânio anterior e ventral pode resultar em defeitos amplos com risco significante de fístula liquórica e/ou exposição da artéria carótida interna. Nestes casos, a reconstrução da base do crânio com a utilização de retalhos vascularizados facilita e encurta o processo cicatrização. O retalho nasoseptal (Hadad-Bassagasteguy Flap) é o nosso método preferido para reconstrução, embora não possa ser utilizado após septectomia posterior ou esfenoidectomias amplas. Foram desenvolvidos na instituição dois retalhos vascularizados na nossa instituição para estes casos: o retalho transpterigóide de fascia temporoparietal (TPFF), para defeitos mais amplos, e o retalho pediculado posterior de corneto inferior (PPITF). O objetivo dete trabalho é descrever nossa experiência com o uso do PPITF.
Métodos.
We developed a flap comprising the inferior turbinate mucoperiosteum pedicled on the inferior turbinate artery, a terminal branch of the posterior lateral nasal artery, which arises from the sphenopalatine artery. We retrospectively reviewed the clinical data of four patients who underwent a skull base reconstruction using a PPITF.
Keywords
Endoscopic surgery, Skull base, CSF leak, Reconstruction, Inferior turbinate, Vascularized flap
Abstract
Introduction: Expanded endonasal approaches (EEA) for the resection of lesions of the anterior and ventral skull base can create large defects with a significant risk of postoperative cerebrospinal leaks and/or exposure of the internal carotid artery. In these cases, a reconstruction using a vascularized flap facilitates rapid and complete healing of the defect. The Hadad-Bassagasteguy (HBF), a posterior pedicle nasoseptal flap, is our preferred reconstructive option; however, a prior posterior septectomy or prior wide sphenoidotomies preclude its use. We have developed two additional pedicled flaps to reconstruct these selected cases: the transpterygoid temporoparietal fascia flap (TPFF), which is suitable for large defects, and the posterior pedicle inferior turbinate flap (PPITF), the subject of this paper.
Methods: We developed a flap comprising the inferior turbinate mucoperiosteum pedicled on the inferior turbinate artery, a terminal branch of the posterior lateral nasal artery, which arises from the sphenopalatine artery. We retrospectively reviewed the clinical data of four patients who underwent a skull base reconstruction using a PPITF.
Results: Four patients underwent a reconstruction with the PPITF after undergoing an EEA that produced a skull base defect associated with a CSF fistula (n=2), an exposed ICA (n=1), or a basilar aneurysm clip (n=1). All patients had undergone posterior septectomies as part of previous EEA's. All flaps healed uneventfully, covering the entire defect.
Conclusion: The PPITF is a viable reconstructive option for patients with skull base defects of a limited size defect and in whom the HBF is not available.
Instituição: 1- Department of Otolaryngology and Head & Neck Surgery 2- Department of Neurological Surgery Minimally Invasive endoNeurosurgery Center, University of Pittsburgh Medical Center
Suporte Financeiro:
Introduction
The indications for endonasal endoscopic skull base approaches (EEA) continue to expand propulsed by technological advances, the acquisition of surgical experience and better understanding of the endoscopic anatomy. As a result, many lesions that previously were resected using traditional craniofacial approaches are currently managed with EEA.1-4
Any reconstruction of the skull base strives to recreate a barrier between the cranial cavity and the sinonasal tract; thus, avoiding a postoperative cerebrospinal fluid leak, decreasing the risk for ascending bacterial meningitis and avoiding the exposure of neurovascular structures.5 Small CSF fistulas can be reconstructed endoscopically with a success rate greater than 95%. This may be achieved using a variety of free grafting techniques without the need for a vascularized flap.6-10 We found, however, that when we apply these techniques to large defects, such as those produced with EEA's, the success rate decreases significantly.11 Despite multiple refinements to optimize these techniques the outcome remained unsatisfactory; thus, prompting the development of vascularized flaps that would promote faster and complete healing. Among the different vascularized flaps, pedicled flaps are most advantageous as they conform to irregular surfaces, and are easier to mobilize into defects than rotation or advancements flaps. These latter flaps require a wide base to recruit an adequate blood supply; henceforth, they are more difficult to mobilize and their base tends to retract toward their original position, away from the defect. We routinely use the Hadad-Bassagasteguy flap (HBF), a nasoseptal pedicled flap based on the nasoseptal artery.12 In patients with prior septectomy or wide sphenoidotomies, however, the HBF blood supply has been interrupted; therefore, it cannot be used. We have developed two additional pedicled flaps that can be used without a craniotomy or maxillofacial osteotomies: the transpterygoid temporoparietal fascia flap (TPPF) and the posterior pedicle inferior turbinate flap (PPITF). The latter is based on the inferior turbinate artery, a terminal branch of the posterior lateral nasal artery (PLNA), which arises from the sphenopalatine artery (SPA).
Others have provided a detailed anatomical description of the arterial supply to the lateral nasal wall.13-17 We provide a brief review of the most important aspects of the vascular supply to the inferior turbinate as it relates to the PPITF. It should be noted, however, that the anatomy of the internal maxillary artery and its branches presents variability and that the nomenclature of the arterial anatomy for this region varies throughout the literature. We should begin at the level of the sphenopalatine artery (SPA), which is one of the terminal branches of the internal maxillary artery. It exits the pterygopalatine fossa (PPF) and enters the nose through the sphenopalatine foramen. It supplies most of the lateral nasal wall including the inferior and middle turbinates through the posterolateral nasal artery (PLNA), also called nasolateral artery. For the design and harvesting of the PPITF, the anatomical course of the PLNA is most relevant. The PNLA runs in a descending vertical or antero-inferior course over the perpendicular plate of the ascending process of the palatine bone. As it courses inferiorly, it gives a substantial branch medially to supply the middle turbinate. It is important to note that in 38% of cases some portion of the LNA runs in an area that is anterior to the posterior wall of the maxillary sinus.15 A descending arterial trunk passes caudally and slightly anterior in a remarkably constant position. It enters the inferior turbinate on the superior aspect of its lateral attachment, 1.0 to 1.5 cm from its posterior tip.17 Some have found that the artery may lie within the bone (50%), within the soft tissue (14%) or following a mixed pattern (36%).16 The artery runs for some distance (mean of 1.2 cm) before piercing the bone and soft tissue and splitting off into two to six branches.16 Others15,16, have suggested two patterns of arterial branching. In the first model the inferior turbinate artery branches at the superior border of the inferior turbinate and divides into two branches, one running anteriorly along the upper portion of the inferior turbinate and another running anteriorly along the midportion of the turbinate. In the second model, the artery divides into two branches that course anteriorly along the upper and mid portions of the inferior turbinate. Both branches remain in a bony canal or are closely applied to the bone for much of the length of the inferior turbinate.15, 16 The lower medial branch gives off arteries, which pierce the bone in its anterior part and form a regular pattern of alternating superior and inferior branches at right angles to the main artery.15 Despite giving off these branches, the artery increases in size as it passes anteriorly, suggesting recruitment of additional blood flow from the angular artery. This latter artery represents another vascular pedicle supplying the inferior turbinate. 18
In this report, we describe our experience with the PPITF, a novel technique for skull base reconstruction.
Material and methods
We retrospectively reviewed the demographic, clinical, surgical, and outcome data of all patients who underwent reconstruction of skull defects using the PPITF, after EEAs that produced CSF leaks or exposure of the internal carotid artery (ICA).
Surgical technique
We decongest the nasal cavity with oxymetazoline 0.05% and infiltrate the lateral nasal wall, just anterior to the inferior turbinate with a solution of lidocaine 1% with epinephrine 1/100,000. We harvest the PPITF after completion of the EEA and ipsilateral to the defect, whenever possible. Bilateral PPITFs may be harvested if necessary. Initially, the inferior turbinate is gently medialized to better expose the entire medial surface of the inferior turbinate and allow visualization of the mucosa from the inferior meatus. The flap may be designed according to the size of the defect but it is best to harvest the entire turbinate to assure adequate coverage. A wider flap may be harvested by extending the lower incision to include the lateral mucoperiosteum of the turbinate and even the middle meatus.
It is best to first identify the SPA as it exits the sphenopalatine foramen and follow it distally to identify the PLNA (FIGURE 1). The flap will be based solely on this vascular pedicle. An outline of the incisions is controlled by direct endoscopic visualization. Two parallel incisions are performed following the sagittal plane of the inferior turbinate, the superior one just above the inferior turbinate (fontanelle of the maxillary sinus) and the inferior one following the caudal margin of the turbinate (FIGURE 1). A vertical incision, placed over the anterior head of the inferior turbinate, connects the two previous incisions. These incisions can be made using endoscopic scissors or preferably an extended insulated needle tip electrocautery. Then, the mucoperiosteum is elevated starting at the anterior aspect of the inferior turbinate. A variable amount of bone may be elevated also, depending on the ease of dissecting the mucoperiosteum from the underlying bone. Care must be taken to avoid injuring the vascular pedicle as it enters at the superior aspect of its lateral attachment, about 1.0 and 1.5 cm from its posterior tip. In addition, it is important to preserve the LNA as it descends vertically over the ascending process of the palatine bone. It may course anterior to the posterior wall of the maxillary sinus; therefore, this should be considered when extending the maxillary antrostomy far posteriorly (i.e. to identify the SPA). Once harvested, the flap is gently unrolled and mobilized to cover the skull base defect.
The PPITF can be applied directly to dura or denuded bone or may be used over a fat graft (FIGURE 2 a & b). It is critical, however, that the vascularized flap be in direct contact with the margins of the defect and to remove any non vascularized tissue between the margins of the defect and the flap. Fibrin glue or other biologic glue is applied over the flap and a sponge nasal packing or the balloon of a 12 Fr. Foley catheter is inserted to press the PPITF against the defect. Care must be taken when inflating the balloon or placing the packing in order to avoid displacement of the flap or compromise of the vascular pedicle. Silicone splints are used to protect the denuded lateral wall and are left in place for about 10 to 14 days.
Results and complications
We used the PPITF in four patients with skull base defects after EEA's that resulted in a communication between the arachnoid space and sinonasal cavity (n=2), exposure of the ICA (n=2), or exposure of the clip used to control a basilar artery aneurysm (n=1) (Table 1). All flaps healed uneventfully covering the entire defect. We have observed no complications resulting from the flap donor site. Crusting, however, occurs and requires frequent debridement until mucosalization is complete. Mucosalization of the inferior turbinate and floor of the nasal fossa was observed after 3 to 4 weeks.
Discussion
One of the most important tenets of reconstruction after skull base surgery is to achieve a complete separation between the cranial cavity and the sinonasal tract.5,19 This goal is critical, whether the surgical approach comprises a conventional craniofacial, endoscopic endonasal, or a combined technique. Our initial technique for skull base reconstruction following EEAs included a monolayer inlay graft to reestablish a tissue barrier. This initial technique reflected our experience with endoscopic reconstruction of CSF leaks. In patients with small skull base defects, free grafting is highly successful, foregoing the need for vascularized flaps. Free grafting of the significantly larger defects produced by EEA, which are comparable in size to those produced by a traditional craniofacial resection, resulted in a high incidence of postoperative CSF leaks.
Our reconstruction technique underwent multiple modifications as a result of the lessons learned with the treatment of our own postoperative CSF leaks.11, 20 First, we adopted the use of a collagen matrix subdural inlay graft to be followed by an acellular dermis onlay graft and then reinforced by free abdominal fat. Then, we adopted the use of a 12 Fr. Foley catheter balloon to support this multilayered reconstruction and to avoid shifting of the grafts. Although these maneuvers reduced our incidence of postoperative CSF leaks after EEA by 50%, it remained at levels that we considered unacceptable.
The advantages of using vascularized flaps for skull base reconstruction after traditional approaches are well established. They promote a complete and rapid healing and accelerate the postoperative recovery, avoiding CSF leaks and/or ascending meningitis. This is critical in clinical situations that involve large defects, especially if the patient has received preoperative radiotherapy or chemoradiotherapy for the skull base lesion. Following this concept, we adopted the use of the HBF; thus, reducing the incidence of postoperative CSF leaks to a level comparable to that of traditional approaches.12 The HBF is a pedicled nasoseptal flap based on the posterior nasoseptal arteries, which are branches of the posterior nasal artery. The HBF is a versatile and reliable flap with a good arc of rotation and large surface area; however, it cannot be used in patients with prior posterior septectomy or large sphenoidotomies. These procedures interrupt the blood supply to the flap precluding its use. We developed two additional pedicled flaps as an alternative to the HBF: the transpterygoid temporoparietal fascial flap (TTPFF) and the PPITF. The TTPFF is based on the anterior branch of the superficial temporal artery. It is a reliable flap that offers excellent area coverage with good arc of rotation and pliability and can be mobilized into the nasal cavity through a transpterygoid tunnel.15, 19 We consider that the TTPFF is an excellent option in cases of large skull base defects. It requires, however, an external incision and an endoscopic nasal transpterygoid approach to create a tunnel that allows its transposition into the nose; thus, it is time consuming
The use of vascularized flaps from the inferior turbinate is not a new concept.21 Anteriorly based flaps have been described for the closure of septal perforations and for the internal lining of nasal defects.18, 21, 22 These mucoperiosteal flaps are based anteriorly on branches from the angular artery, a branch from the facial artery.18 The main inferior turbinate bloody supply, however, comes from the inferior turbinate artery, a terminal branch from the LNA, which is a terminal branch from the SPA.13 The inferior turbinate artery enters the turbinate posteriorly in its lateral surface.15 We developed the PPITF for skull base reconstruction based on this artery, which provides a reliable vascular pedicle with good arc of rotation that reaches most of the ventro-caudal skull base defects. The PPITF surface area is not as extensive as that of the HBF or the TTPFF. According to previous anatomical analysis of the anteriorly based inferior turbinate flap, the mucosal surface area of this flap is about 4.97 cm2 (2.8 cm length with 1.7 cm width). 18 Bilateral flaps, however, can be harvested to cover larger defects.
One disadvantage of using the PPITF is the formation of crusting over the inferior turbinate in the postoperative period. In our three cases mucosalization of the donor site was observed after a period of 3 to 4 weeks. In addition, in patients with a prior inferior turbinectomy or atrophy of the turbinate, the mucosal surface may have inadequate size; therefore, these are relative contraindications to the PPITF.
Conclusion
The PPITF is a viable option for the reconstruction of moderate size skull base defects in patients with a prior posterior septectomy. Its use may be expanded to the repair of other lesions such as nasal septal perforations, oro-nasal and oro-antral fistulas.
Table 1.
|
Age |
Gender |
Pathology |
Site of defect |
Major vessel exposure |
CSF leak |
Followup (months) |
|
51 |
Female |
Chordoma |
Clivus |
No |
Yes |
5 |
|
55 |
Male |
Chordoma |
Clivus/parasellar |
Yes |
Yes |
6 |
|
48 |
Female |
Pituitary macroadenoma |
Sella/parasellar |
Yes |
No |
11 |
|
57 |
Male |
Basilar artery aneurysm |
Clivus |
Yes*
|
No |
2 |
* aneurysm clip
References
Figura 1.

Drawing of the left lateral nasal wall outlining the incisions. A wide naso-antral window (shaded area) was opened to facilitate the identification of the sphenopalatine artery (SPA) and the posterior lateral nasal artery (PLNA).
Figura 2A.
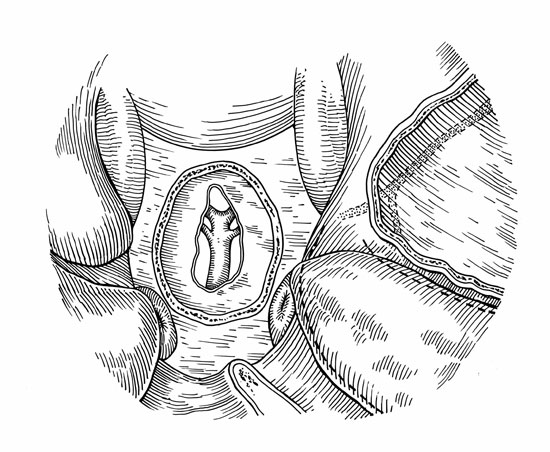
a) Drawing of a clival defect
Figura 2B
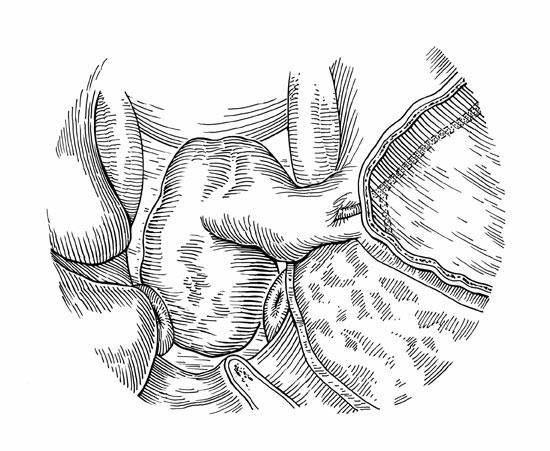
b) Drawing of the PPITF covering the defect
Figura 2C.
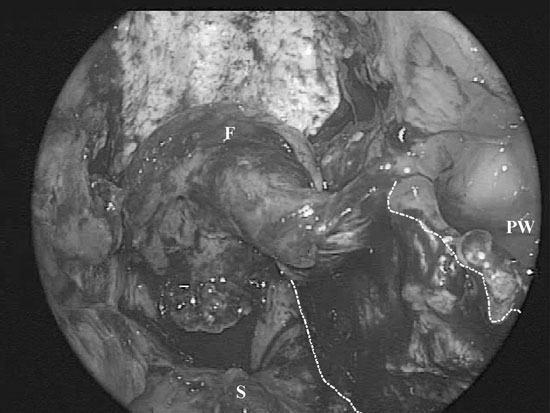
c) Intraoperative photograph of the repair. A black line has been added to highlight the edges of the flap.